Disease area
Multiple sclerosis (MS) is an autoimmune disease in the central nervous system (CNS) where the chronic inflammation trigger and promote neuro-axonal degeneration. A majority of patients are diagnosed at age 20 to 40 and it is one of the common causes of disability for young adults. The etiology of the disease is unknown, but considerable evidence indicates that interplay between genetic background and environmental factors play an important role. Blocking acute inflammation using disease modifying drugs and symptoms relieving treatment are current strategies used in clinical practice for patients with MS, unfortunately, there is no cure for MS at present.
Rationale
Historically MS has been considered a T-cell mediated disorder [1], however increasing evidence from past decades suggest that B cells are not only a homogeneous population as antibody producing cells, but also an important player with antibodyindependent function, such as antigen presenting and cytokine production. Observational studies using selective B cell depletion therapy (anti-CD20 antibodies) showed highly beneficial effects in limiting new MS disease activity [2,3] without obvious total serum IgG level reduction, but rather decreasing of several proinflammatory mediators such as IL-6 and TNF-α, which indicate that the beneficial effects of B cell depletion therapies attribute to the antibody-independent role of B cells.
Aim
To investigate cytokine production of purified primary B cells and cell viability upon stimulation in presence of well-defined chemical probes and compounds.
Methods
General protocol: CD19+ B cells purified from Peripheral Blood Mononuclear Cells (PBMCs) are cultured for 2 days with presence of stimulation cocktail and compounds. The effect on cytokines secretion from stimulated B cell upon addition of chemical probes is investigated by multiplex beads array in cell supernatants and cell health status is measured by CellTiter-Glo assay.
Cell culture condition: Cells are cultured in 384 well plate at 100 000 cells/ml, 50µl/well, in duplicates. Culture medium is RPMI supplemented with 10% heat inactivated bovine serum. Chemical probes (at final concentration 0,1 µM or 1 µM) are preloaded to plate at start of culture and pre-treated for 30 min before stimulation cocktail added. Controls include unstimulated condition (cell in culture medium without added cytokines), stimulated condition with vehicle control only (0,1% DMSO), stimulated condition with the Bruton’s tyrosine kinase inhibitor ibrutinib in 0.1% DMSO.
Readout: Flow cytometry is done on freshly isolated B cells to determine the purity and the phenotype of different B cell subsets. Cytokines secretion was measured using multiplex-bead-array.
Results
A set of 180 probes were tested in 4 blood donors including 1 patient with MS and 3 healthy donors. The effect on cytokine secretion of B cells on day 2 of cell culture in presence of B cell stimulating agents are expressed as fold change normalized to the vehicle control (0,1% DMSO).
In all donors, the positive control, ibrutinib decrease IL6/TNF-α/GM-CSF secretion more than 50% without affecting cell viability (Figure 1A), level of IL-Iβ is low and highly variable in the four donors tested. Out of 180 screened chemical probes, ~20 compounds showed some level of reduction of IL-6, TNF-α and/or GM-CSF, without compromising cell health status (Figure 1B). Bromodomain inhibitors and kinase inhibitors dominate among those leading hits that inhibit cytokine secretion.
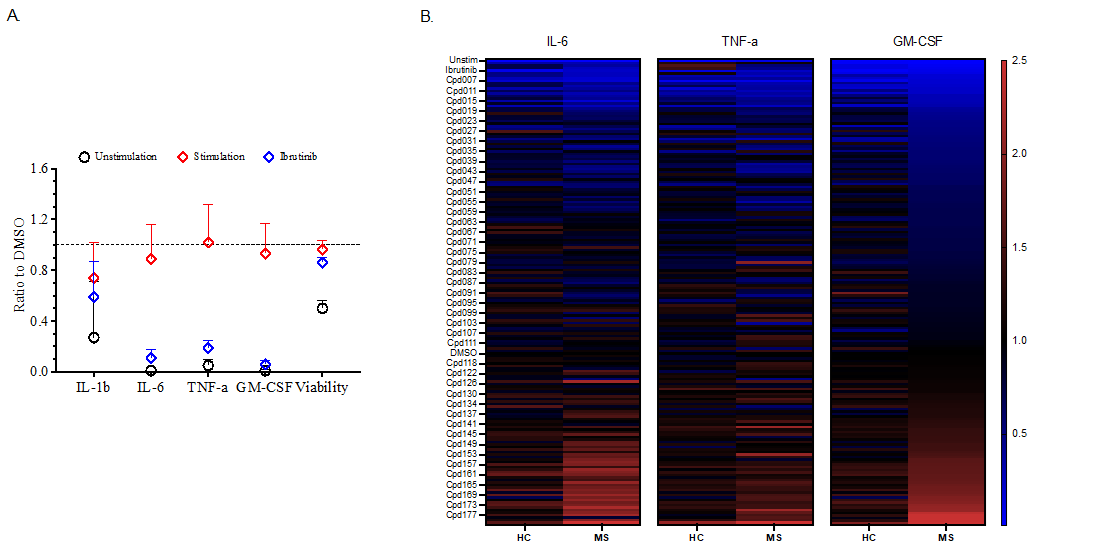
Figure 1: Preliminary screening results from healthy donors (n=3) and Multiple Sclerosis patient (n=1). A) Experimental control includes medium (unstimulation), stimulation cocktail and positive control Ibrutinib. B) Fold change normalized to DMSO control for all the compounds screened on cytokines’ production.
Conclusions
The preliminary screening results of chemical probes on cytokines production in the isolated B cell assay look promising. Additional healthy controls and MS patients will be added with the final release of the data set.
References
- Dendrou, C. A., Fugger, L., & Friese, M. A. (2015). Immunopathology of multiple sclerosis. Nat. Rev. Immunol., 15(9), 545–558
- Bar-Or, A., et al. (2008). Rituximab in relapsing-remitting multiple sclerosis: a 72-week, open-label, phase I trial. Ann. Neurol., 63(3), 395–400
- Li, R., Patterson, K. R., & Bar-Or, A. (2018). Reassessing B cell contributions in multiple sclerosis. Nat. Immunol., 19(7), 696–707.



